What is pharmacogenetics?
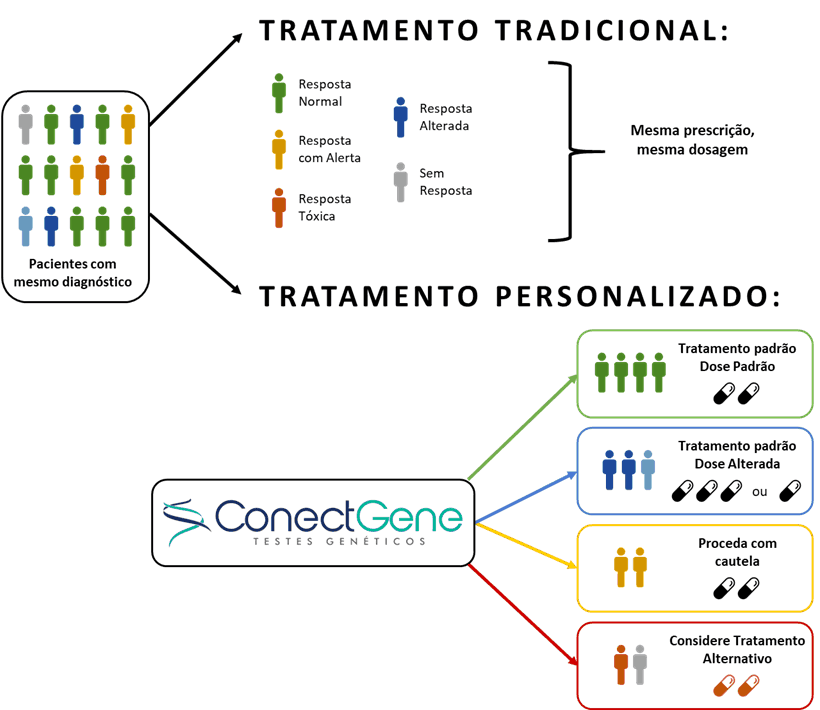
Pharmacogenetics (or pharmacogenomics) aims to predict how individual genetic variability impacts drug absorption and metabolism. While a treatment may work well for one individual, the same treatment may be ineffective or cause adverse side effects in another patient.1,2
The cost of drug side effects
Drug-related adverse events have a serious impact on health
The incidence rate of drug-related adverse events in the US patient population is estimated at 6.7%3
It is estimated that the costs associated with these adverse effects exceed U$ 100B
Hospitalization rates for patients using warfarin would be reduced by 31% if patients had pharmacogenetic testing
Drug absorption
Membrane transporters are crucial determinants in the absorption of drugs. The family of organic anion-transporting polypeptides is especially important in mediating the absorption of many drugs by hepatocytes. Genetic variability in the genes encoding these transporters is associated with significant differences in the pharmacokinetics of drug absorption. For example, a single nucleotide polymorphism in the SLCO1B1 gene (encodes OATP1B1) can lead to impaired absorption of many statins, including simvastatin, pitavastatin, atorvastatin and rosuvastatin7. In addition, a high plasma concentration of simvastatin is also associated with a higher risk of drug-induced myopathy
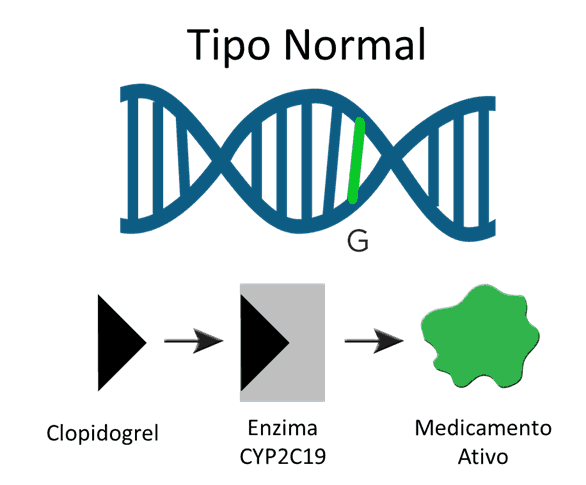
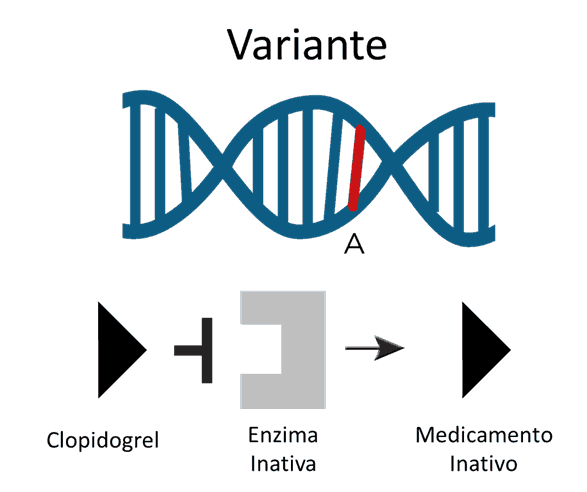
The phenotypes of metabolizers
Patients who have genetic variants associated with rapid drug metabolism (ultra-rapid metabolizers) may benefit from high doses to achieve the desired therapeutic response. On the other hand, patients with variants that cause slow metabolization of the drug are at risk of intoxication and are twice as likely to have adverse side effects. For example, three different variations of CYP2C19 are correlated with a reduced ability to metabolize the platelet antiaggregant clopidogrel and carriers have a higher risk of cardiovascular difficulties. A recent study focusing on one of the three variants (CYP2C19*2) showed that carriers were twice as likely to suffer cardiovascular ischemia or death within a year of taking the drug. In response to the evidence from the studies, the FDA (Food and Drug Administration – USA) is now recommending that doctors consider an alternative treatment for patients in the slow metabolizer category.
The higher incidence of toxicity in slow metabolizers has also been observed in several antidepressants, including desipramine, venlafaxine, amitriptyline and haloperidol. In addition, a specific CYP2D6 polymorphism can inhibit the analgesic effect of opioids such as codeine, tramadol and oxycodone. Conversely, ultra-fast metabolizers can suffer from lethal levels of toxicity from the same opioids.
Additional Sources
Still have questions?
Send your questions to:
Bibliographical references
- Kitzmiller, J. P., Groen, D. K., Phelps, M. A. & Sadee, W. Pharmacogenomic testing: relevance in medical practice: why drugs work in some patients but not in others.Cleveland Clinic journal of medicine 78, 243-257, doi:10.3949/ccjm.78a.10145 (2011).
- Wang, L., McLeod, H. L. & Weinshilboum, R. M. Genomics and drug response.The New England journal of medicine 364, 1144-1153, doi:10.1056/NEJMra1010600 (2011).
- Samer, C. F., Lorenzini, K. I., Rollason, V., Daali, Y. & Desmeules, J. A. Applications of CYP450 testing in the clinical setting.Molecular diagnosis & therapy 17, 165-184, doi:10.1007/s40291-013-0028-5 (2013).
- Lazarou, J., Pomeranz, B. H. & Corey, P. N. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies.JAMA: the journal of the American Medical Association 279, 1200-1205 (1998).
- Van Driest, S. L.et al.Clinically actionable genotypes among 10,000 patients with preemptive pharmacogenomic testing.Clinical pharmacology and therapeutics 95, 423-431, doi:10.1038/clpt.2013.229 (2014).
- Epstein, R. S.et al.Warfarin genotyping reduces hospitalization rates results from the MM-WES (Medco-Mayo Warfarin Effectiveness study).Journal of the American College of Cardiology 55, 2804-2812, doi:10.1016/j.jacc.2010.03.009 (2010).
- Kalliokoski, A. & Niemi, M. Impact of OATP transporters on pharmacokinetics.British journal of pharmacology 158, 693-705, doi:10.1111/j.1476-5381.2009.00430.x (2009).
- Group, S. C.et al.SLCO1B1 variants and statin-induced myopathy-a genomewide study.The New England journal of medicine 359, 789-799, doi:10.1056/NEJMoa0801936 (2008).
- Shuldiner, A. R.et al.Association of cytochrome P450 2C19 genotype with the antiplatelet effect and clinical efficacy of clopidogrel therapy.JAMA : the journal of the American Medical Association 302, 849-857, doi:10.1001/jama.2009.1232 (2009).
- Rost, S.et al.Mutations in VKORC1 cause warfarin resistance and multiple coagulation factor deficiency type 2.Nature 427, 537-541, doi:10.1038/nature02214 (2004).
- Johnson, J. A.et al.Clinical Pharmacogenetics Implementation Consortium Guidelines for CYP2C9 and VKORC1 genotypes and warfarin dosing.Clinical pharmacology and therapeutics 90, 625-629, doi:10.1038/clpt.2011.185 (2011).